Misinformation continues to take hold in the plastics industry on how to determine the amount of moisture in your resin. It’s time once again to set the record straight.
July 28, 2010
Misinformation continues to take hold in the plastics industry on how to determine the amount of moisture in your resin. It’s time once again to set the record straight.
At last year’s combined NPE and Antec, I had the pleasure of sitting on a panel in the Failure Analysis and Prevention Special Interest Group sessions with Dr. Myer Ezrin. “Mike,” as he is known to his colleagues, is a rare combination of university scholar and practical problem solver. He has managed to keep one foot in the world of education, much to the benefit of those who have had an opportunity to study under him, and the other foot in the world of industry, where problems must be solved every day with practical approaches that constantly test the theories taught in our universities.
|
During the session, the subject of moisture analysis came up. I was sitting next to Dr. Ezrin and while another member of the panel answered the question that had been posed, Mike turned to me and said, “They’re talking about Karl Fischer.” It was a statement so matter-of-fact, that he could have easily appended the phrase, “of course.”
But the questioner was not talking about Karl Fischer titration. Instead, he was referring to the more commonly used loss-on-drying (LOD) or loss-in-weight systems that are widely in use in the plastics industry today. In our sidebar conversation I explained the loss-in-weight technique and the fact that the vast majority of molders who test for moisture content in their resins use this method. Mike’s response was as genuine as it was correct. “You can’t do that.” Mike’s concern pertained to how the test distinguishes between moisture and all of the other compounds that can be evolved from a polymer when it is heated.
One of the techniques that Mike has specialized in over the years to solve problems is known as gas chromatography-mass spectroscopy (GC-MS). This method involves heating a substance to extract volatile compounds. These compounds are then injected into a column that separates them and identifies them according to the time it takes to move through the column and their molecular weight. Related techniques that achieve similar objectives are known as desorption-mass spectroscopy (DMS) and pyrolysis-mass spectroscopy (PY-MS). Anyone who has performed these tests as often as Dr. Ezrin has is fully aware of the fact that when you heat a plastic material, you evolve a very large number of compounds.
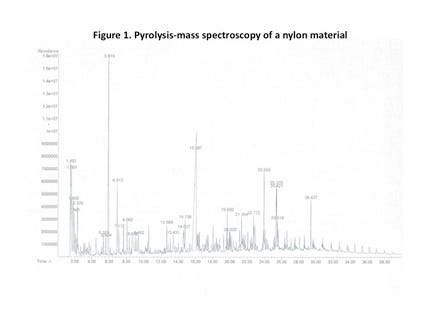
Figure 1 shows the results of a PY-MS test performed on a nylon material. Each peak in the graph represents a compound that was evolved from the material. One of these will be moisture (water) and the rest are something else. Because water is one of the lower-molecular-weight compounds that will be contained in a polymer, it will be one of the first to evolve; therefore, it is likely to be one of the peaks captured in the first 1-2 minutes of the scan. The peak heights represent relative abundance and it is fairly easy to see that when the test time approaches 30 minutes, most of the volatile material has been captured. It is equally easy to see that the single peak that represents water accounts for only a small percentage of the total material that is collected.
|
This result serves to illustrate the reason for Dr. Ezrin’s incredulity. When so many potentially volatile compounds are contained in a plastic material, how can one possibly quantify the water content of the material by simply heating a sample, measuring the resulting weight loss, and concluding that the entire weight loss is water? The answer is simple: You can’t. So why is it that almost every molder in the industry that is measuring moisture is using an LOD system?
To understand this we need to look at the various methods by which moisture content is determined and how the plastics industry has grown up with these methods. Material suppliers, with excellent lab facilities and a highly skilled staff, have traditionally measured moisture content using the method that is the gold standard in the industry, Karl Fischer titration. This method, named for its inventor, was developed in 1935. It operates on the principle that water will produce a unique chemical reaction when introduced into a particular mixture of chemicals. We will get into more details regarding this method in Part 2 of this article next month.
No DIY testing
Karl Fischer titration is not the only method for measuring moisture. Several other chemical methods have been developed over the years that meet the demands of industries where the requirements for precision and accuracy are even greater than those of the plastics industry. However, they are all based on monitoring a reaction between water and some chemical or combination of chemicals. These reactions only occur with water. Some type of sensor must then be used to measure the effects of the water on the other chemicals or make a quantitative measurement of a product formed by the chemical reaction. All this is best accomplished in a laboratory environment and has typically not been something easily adapted to a manufacturing arena.
With the introduction of the microprocessor, monitoring many of these types of reactions and translating them into an easily understood result has been placed within the grasp of those in the world of manufacturing. Karl Fischer titration is among those techniques that has benefited from this revolution. However, it is still a laboratory technique that involves chemicals and glassware and therefore must be run in a controlled environment; it is not a process for the factory floor. Because the device looks intimidating and unfamiliar to most people with a manufacturing background, it has not achieved significant penetration among plastics processors despite its ability to make unambiguous measurements of moisture content.
Because of this perceived degree of difficulty, and some exaggerations about the technique from those marketing competitive devices, the processing industry has overwhelmingly opted for LOD systems. These systems are much simpler. They essentially consist of a heating device and a very sensitive balance. A sample is weighed and then heated at a prescribed temperature. The amount of lost mass that occurs during the heating routine is considered to represent the moisture content of the material being tested.
LOD instruments have their place. They are useful for products where the amount of moisture being measured represents a relatively large percentage of the sample and where the amount of other volatile compounds present in the sample represents a small fraction of the material that can be evolved at elevated temperatures. Foodstuffs and pharmaceutical products are good examples. Some food products can hold up to 15% moisture by weight and the amount of other volatiles often totals no more than 0.1%-0.2% of the product. In cases like this, LOD systems work fine. They produce the desired accuracy and there is little chance that other compounds will contribute enough to the weight loss to upset the precision of the measurement.
Unfortunately, plastic materials do not fall into this category. In a situation where moisture measurement is being performed to determine suitability for processing, the highest value that would represent an acceptable moisture content would be 0.2% (2000 ppm) for unfilled nylon materials. For many of the most critical polymers such as polycarbonate and PET polyester, the maximum allowable moisture content is 0.02% (200 ppm). To measure moisture content with the appropriate precision, a suitable instrument should be able to provide resolution to one-tenth of the value being measured, or in this case 20 ppm. In addition, the material should not contain more than a few parts per million of other volatile materials since these will contribute to the weight loss and be erroneously identified as moisture.
However, as the result in Figure 1 shows, these conditions are not met in plastic materials. The amount of other volatile compounds in a commercial molding material far exceeds the amount of moisture that is absorbed by the material. The compounds other than water that are lost as the result of simple heating such as is performed in an LOD system include additives and residuals. The additives may be some combination of slip agents, antistats, plasticizers, antioxidants, UV stabilizers, dyes and organic pigments, and even some flame-retardants. Residuals include low-molecular-weight polymer fragments such as monomer, catalyst residues, and other so-called “cats and dogs” that are not completely removed during the pelletizing and compounding processes.
Not all of these will be evolved when a material is heated to a temperature high enough to remove all the moisture from a sample. But the amount that will be removed will be much greater than the amount of water, even if the sample is quite wet. If it is dry, the task of distinguishing moisture from all the other compounds with peaks as shown in Figure 1 is hopeless. This is the reason for Dr. Ezrin’s pronouncement that “you can’t do that.”
Misinformation from 
trusted sources
So why does the processing industry ignore such guidance from the most knowledgeable in the industry? In part because we don’t know any better. Molders are not polymer chemists and so they are largely unaware of the potential for other substances in their materials to interfere with an accurate determination of moisture content. Another reason is that the processing industry is encouraged to embrace LOD by the manufacturers of these instruments. They advertise ease of use and they tell horror stories of how difficult it is to use the instruments that are designed to be moisture specific.
A particularly troubling trend is the fact that representatives of some resin suppliers have reportedly begun to endorse these systems as a valid tool for measuring moisture content in plastic materials. But ignorance does not banish the laws of chemistry and ease of use does not qualify a technique as scientifically sound.
Here are a few of the statements that I have heard regarding moisture analysis:
• Karl Fischer titration uses expensive chemicals that are toxic.
• Karl Fischer equipment includes glassware that can break.
• Karl Fischer titration is too difficult for the average manufacturing person to master.
• LOD systems are calibrated to produce moisture measurements that are as precise as those obtained using the more sophisticated lab equipment.
• Ongoing calibration of loss-on-drying systems is not necessary.
In fact, the science of measuring moisture is not simple, but it can be learned by those involved in molding plastic components. In Part 2 we will review the differences between moisture-specific measurement techniques and the simpler methods the industry embraces. We will also look at the fact and fiction behind the statements above and define the requirements for a fundamentally sound moisture measurement system. Given the fact that often the difference between a good and bad part is the resin’s moisture content, the stakes are too high to rely on guesswork.
About the Author(s)
You May Also Like