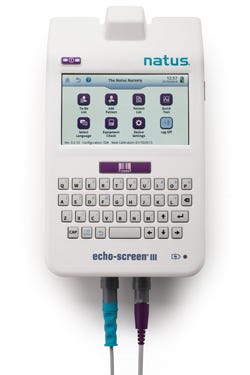
A medical device used to screen the hearing of newborns has undergone a material makeover. Components of the Echo-Screen, which has been on the market for 10 years, originally were fabricated from a PC/ABS alloy. Presumably because of the BPA controversy, which some studies have linked to human health concerns, Natus Medical (San Carlos, CA) established new material requirements for its infant-care products, starting with the Echo-Screen. The company chose Tritan copolyester MXF121 from Eastman Chemical Co.
September 18, 2014
A medical device used to screen the hearing of newborns has undergone a material makeover. Components of the Echo-Screen, which has been on the market for 10 years, originally were fabricated from a PC/ABS alloy. Presumably because of the BPA controversy, which some studies have linked to human health concerns, Natus Medical (San Carlos, CA) established new material requirements for its infant-care products, starting with the Echo-Screen. The company chose Tritan copolyester MXF121 from Eastman Chemical Co. (Kingsport, TN) as the material of construction for its new Echo-Screen III device.
The opaque Tritan MXF121 copolyester is not manufactured with BPA and has superior chemical resistance, says Eastman, and the material also is made without halogens or ortho-phthalate plasticizers. Introduced earlier this year for the medical market, the material is intended for use in handheld and other medical device housings. Its chemical resistance is an especially attractive feature for these applications.
 Medical-device housings increasingly must withstand exposure to aggressive cleansers and disinfectants, as healthcare environments try to prevent the spread of hospital-acquired infections, says Ellen Turner, Market Development Manager, Speciality Plastics, Medical Devices, Eastman. "Eastman Tritan copolyester MXF121 provides excellent chemical resistance against these cleaners and disinfectants. This, as well as the material's toughness, can translate into reduced device failure and greater peace of mind for healthcare professionals and patients," she adds.
Medical-device housings increasingly must withstand exposure to aggressive cleansers and disinfectants, as healthcare environments try to prevent the spread of hospital-acquired infections, says Ellen Turner, Market Development Manager, Speciality Plastics, Medical Devices, Eastman. "Eastman Tritan copolyester MXF121 provides excellent chemical resistance against these cleaners and disinfectants. This, as well as the material's toughness, can translate into reduced device failure and greater peace of mind for healthcare professionals and patients," she adds.
Natus Medical also chose the material for its mechanical properties, impact resistance, aesthetics, and moldability, according to a press release distributed by Eastman. Natus used existing production molds with only minor modifications, because the linear mold shrinkage of Tritan MXF121 is nearly identical to PC/ABS.
"The Natus development team was able to meet user requirements for a DEHP-free and BPA-free product by partnering with Eastman on material selection," said Jim Hawkins, President and CEO of Natus Medical. "We believe this next-generation product will be very well-received by our international customers as hearing screening adoption continues to grow worldwide."
The Echo-Screen III device, a compact, all-in-one product that is used primarily to screen newborns' hearing, was introduced outside the United States earlier this year, and received FDA clearance in August.
Eastman and Natus say they plan to work together to convert additional product lines to Eastman's Tritan copolyester MXF121.
About the Author(s)
You May Also Like




