The contract manufacturer and partner of medtech OEMs is building a medical manufacturing plant in Poland, expanding production capacity in China, and upgrading a facility in Arkansas.
October 20, 2021
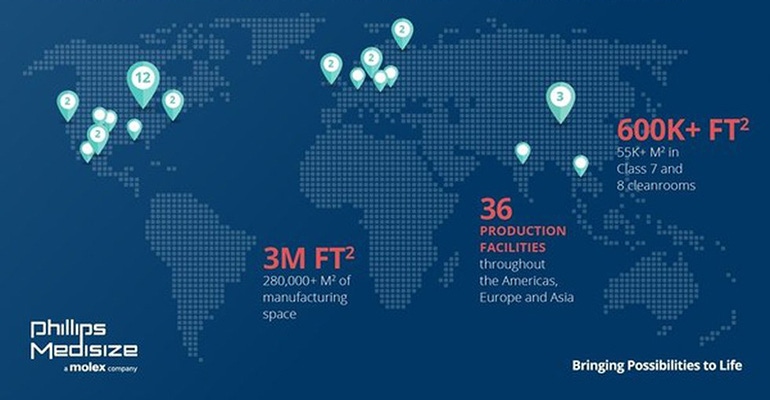
“Think globally, act locally” may be an old saw, but in the context of today’s supply-chain chaos, it retains timeless business wisdom. That came to mind as I was reading an announcement from medtech contract manufacturer Phillips-Medisize about an expansion of its global manufacturing footprint. The company’s global reach currently encompasses 36 production plants, and while offshoring is part of the business model, it’s also true that this constellation of facilities serves the mission of meeting localized demand.
Commenting on the company’s focus on providing customers with end-to-end capabilities wherever they may be located, Paul Chaffin, President, Medical and Pharmaceutical Solutions, at Phillips-Medisize's parent company Molex, said: “As the preferred partner for leading global customers, we continually invest in new talent and technologies that increase the capacity and capabilities of Phillips-Medisize across the entire value chain. Our expanded global reach and resources will enable us to solve complex development and manufacturing challenges while meeting escalating customer demands for more localized production, supply-chain management, and accelerated go-to-market strategies.”
On that note, Phillips-Medisize is building a state-of-the-art medical manufacturing facility in Katowice, Poland. Slated to open in 2022, the site will complement production sites and innovation centers in Asia, Europe, India, Mexico, and North America, said the announcement. Phillips-Medisize also is expanding production capacity in Suzhou, China, to serve both global and regional pharmaceutical and medtech customers. Here in the United States, an existing Molex production facility in Little Rock, AR, is being renovated to enable Phillips-Medisize to “keep pace with ever-increasing [domestic] requirements for high-volume, diagnostic device manufacturing.”
Once these expansions are complete, Phillips-Medisize said that it will offer nearly three million square feet (280,000 square meters) of country- and region-specific manufacturing space and R&D capabilities worldwide. The company also will support 600,000 square feet/55,000 square meters of Class 7 and 8 cleanrooms, which complement existing tool-building sites as well as global quality and regulatory systems. In 2020, Phillips-Medisize completed a new facility in St. Croix Meadows, WI, with 285,000 square feet (26,000 square meters) supporting production of molded components for medical diagnostic customers. That facility also includes a nearly 64,000-square-foot (6,000-square-meter) ISO 14644-1–certified Class 8 cleanroom.
A self-described one-stop shop for medical device manufacturing, Phillips-Medisize focuses on design for manufacturability and assembly expertise, it said in the announcement. Expanded capabilities in complex molding, drug and reagent handling, as well as final packaging and serialization, enable customers to consolidate global supply chains while optimizing go-to-market strategies, the company added.
Phillips-Medisize’s combined expertise in plastics, metals, electronics, and connectivity facilitate the design and manufacture of combination devices, such as needle-based injection systems and wearable injection technologies.
Phillips-Medisize was acquired by Molex in 2016, which brought its electronics expertise and global reach into the fold. At the time of the acquisition, Matt Jennings, Chairman, President, and CEO of Phillips-Medisize, said, “Molex’s global scale in electronics, coupled with Phillips-Medisize’s strength in designing and manufacturing innovative products for medical device customers, will help us become a global leader in connected health solutions.”
Indeed, in the press release published yesterday, Phillips-Medisize announced a strategic initiative with Credence MedSystems. The agreement will support scaling of the manufacture of the Credence Companion and Dual Chamber Reconstitution Systems. Other potential collaboration areas include autoinjectors and connectivity products, said Credence MedSystems. The Credence Connect Auto-Sensing Injection System is a connected device for syringes targeting chronic disease management and enabling improved clinical trial compliance. “We’re excited to leverage Phillips-Medisize’s world-class manufacturing as we scale toward high-volume automation at the new state-of-the-art facility in Poland,” said Jeff Tillack, COO at Credence MedSystems. “The opportunity to scale production in close proximity to our operations and European customers will accelerate market delivery of our innovative solutions to meet the needs of our pharma customers and their end-users.”
Other recent deals include a design and development program with GlucoModicum for the rapid scale-up of manufacturing for its Talisman needle-free continuous glucose monitor and an agreement with Paris-based Eyevensys to optimize the design, development, and high-volume manufacturing of its ocular device component that powers the delivery of novel gene therapies for treating various eye diseases.
On average, Phillips-Medisize said that it commercializes 50 new products a year for customers, including the first-to-market, FDA-registered drug-delivery device utilizing a connected health system.
About the Author(s)
You May Also Like




