Because it is biocompatible and withstands high temperatures, and has a number of desirable properties such as flexibility, transparency, and compatibility with thin-wall molding, liquid silicone rubber (LSR) is increasingly specified for medical applications. The material is used in catheters, tubing, and sealing caps, and the growing homecare sector will further fuel demand for the material.
March 10, 2014
Because it is biocompatible and withstands high temperatures, and has a number of desirable properties such as flexibility, transparency, and compatibility with thin-wall molding, liquid silicone rubber (LSR) is increasingly specified for medical applications. The material is used in catheters, tubing, and sealing caps, and the growing homecare sector will further fuel demand for the material. LSR is not a simple material to process, however. The molder must match a traditional two-part LSR with a fixed cure rate to the part geometry, size, and weight and to the injection molding equipment that will be used. Bluestar Silicones has developed a process that eliminates the conventional Part A/Part B LSR system, giving the molder better control of cure reactivity, and, thus, of the overall molding process. Reduced cycle times and less scrap are among the reported advantages of the LSR Select system. Umar Latif, Technical Services Manager, explained the LSR Select concept at the Molding 2014 conference in Newport Beach, CA, last week.
The typical two-drum LSR process, in which the base chemistry is laced with a catalyst in Part A and a crosslinker and inhibitor in Part B, leaves few options for the molder to optimize the cure profile, said Latif. Essentially, he or she can adjust the mold temperature or choose a different LSR product. Getting the right balance can translate into many hours of material and process selection, and require an extensive inventory of grades of silicone to meet the tooling requirements for a portfolio of parts. Once production begins, the molder also must deal with lot-to-lot variation that can affect viscosity, cure reactivity, and mechanical properties. During the liquid injection molding process, the molder typically adjusts injection speed, temperature, and other parameters to produce a part that meets end-use requirements.
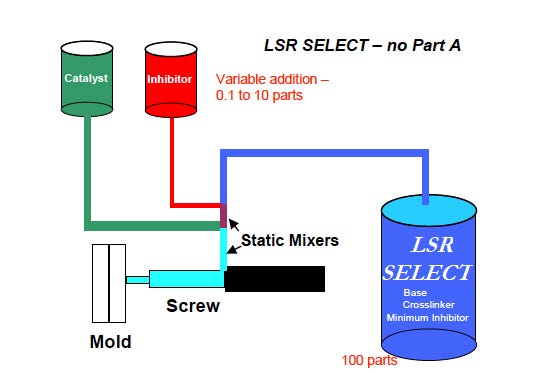
LSR Select replaces the conventional Part A and Part B kit with a base component that contains vinyl functional polymers, hydride crosslinkers, and a small quantity of inhibitor. The material's cure reactivity is adjusted by adding the appropriate amount of LSR Select inhibitor. "The molder now has control over the silicone chemistry to optimize cure," said Latif.
During his presentation, Latif outlined a field study that Bluestar Silicones conducted. The base product was pumped into molding equipment using a standard 1:1 mix ratio meter-mix pumping system. The inhibitor and catalyst were introduced into the LSR base stream and travelled through a static mixer into the injection unit. Both hot and cold runner systems were used and parts ranged in size from 11.5 to 422 grams. The result? Cycle times were reduced by 19 to 50% compared with conventional LSR molding, according to the company.
To further optimize the process, Bluestar Silicones has partnered with Graco Inc. to develop a production-ready closed-loop metering system that provides elevated levels of repeatability and traceability. An intuitive touch screen interface affords the user precise control over cure kinetics of the parts, according to the companies.
In addition to a significant reduction in cycle times, the LSR Select concept allows molders to reduce product line complexity, since the same catalyst and inhibitors are used for all LSR Select durometers, says Bluestar Silicones. An optional third-stream injector enables the introduction of additives, further reducing the need to stock specialized LSR products.
About the Author(s)
You May Also Like




