Despite its many benefits including patient convenience and cost reduction, point-of-care (POC) diagnostics has not achieved the commercial potential that many expected. That will change in the coming years, according to Yole Développement (Lyon, France), which predicts that the sector will reach $38 billion by 2017, representing 16% of the in vitro diagnostics market. That is welcome news for patients and payers.
February 19, 2014
Despite its many benefits including patient convenience and cost reduction, point-of-care (POC) diagnostics has not achieved the commercial potential that many expected. That will change in the coming years, according to Yole Développement (Lyon, France), which predicts that the sector will reach $38 billion by 2017, representing 16% of the in vitro diagnostics market. That is welcome news for patients and payers. It's also good news for polymer suppliers and plastics processors, since plastic is becoming the reference substrate of POC applications, according to Yole Développement.
Polymers, glass, silicon, metal, and ceramics typically are used to manufacture microfluidic devices. The material of choice depends on the application, says Dr. Benjamin Roussel, an analyst at Yole Développement.
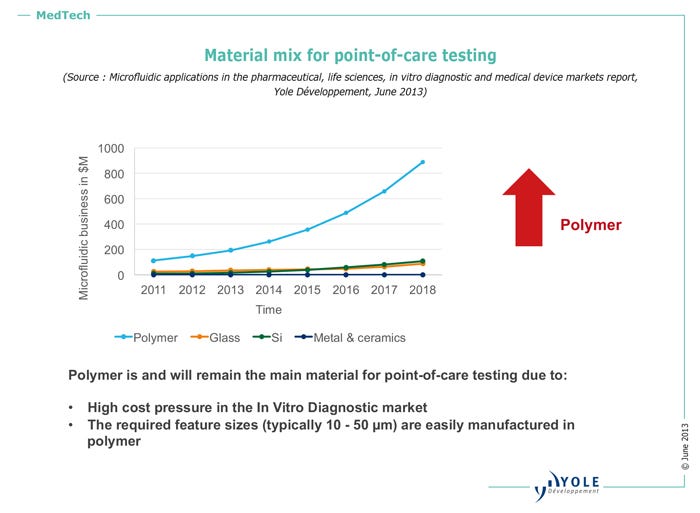
"Plastics are desirable when the device has [relatively] large channels," Roussel told PlasticsToday. "The main criteria are cost and disposability, because of contamination concerns. Plastics currently have 90% market share for POC devices, and we project that its share will continue to rise, mostly because of cost pressure." Microfluidics-based POC testing will account for more than $16 billion by 2017, he adds.
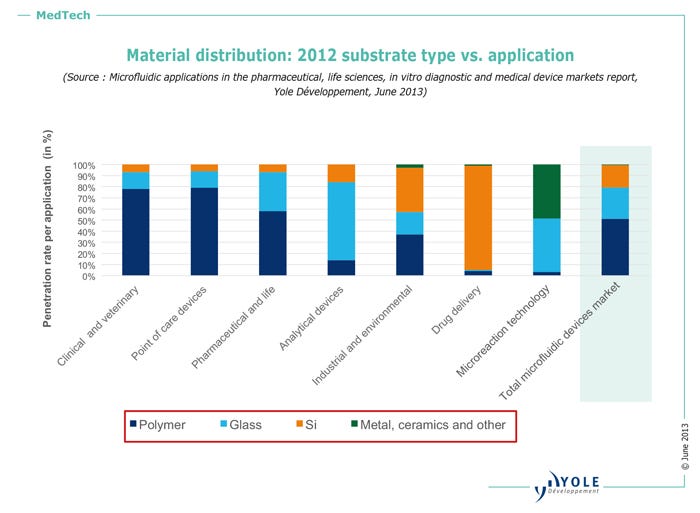
Because of their relatively low price points and properties, poly methyl methacrylate (PMMA) and cyclic olefin copolymer (COC) are the preferred materials for POC applications. PMMA gets the nod for research applications, says Roussel, because of cost and manufacturability. For industrial uses, COC is preferred, owing to its optical properties. In fact, COC is used in approximately 80% of POC applications.
A report published by Yole Développement provides in-depth analysis of the microfluidics-based POC market. It explores the technical, regulatory, and economic hurdles that this sector must—and will—overcome. The many applications in what Yole calls a highly segmented market are analyzed, and the top 10 companies are profiled. The report is available through the ResearchAndMarkets website.
About the Author(s)
You May Also Like




