June 13, 2022
Global medical technology company BD (Becton, Dickinson, and Co.) and Mitsubishi Gas Chemical Co. Inc. (MCG) recently signed a letter of intent to discuss a partnership to explore new ways to advance prefillable syringe technology for biologic drug delivery. Specifically, the agreement states that they will investigate further development of Oxycapt, an innovation from MGC that integrates what it calls the best of plastic and glass for plastic syringes.
Together, the companies will look into applying Oxycapt technology to the next generation of prefillable syringes for advanced biologic pharmaceuticals.
BD said in the announcement that it has been working for decades to optimize the performance of prefillable syringes, which improves the delivery of drugs and vaccines. It is the world leader in supplying prefillable syringes to the biopharmaceutical industry, with more than 70% of the top 100 biopharmaceutical companies reportedly using its devices. BD manufactures both glass and plastic syringes to meet the unique needs of drugs.
“The biopharmaceutical industry continues to rely on prefillable syringes for the effective, reliable, and consistent administration of medications,” said Bruno Baney, Vice President, R&D, BD Medical – Pharmaceutical Systems. “As our customers continue to grow their drug pipelines, we are constantly exploring solutions to deliver newer compounds in robust, stable, and ready-to-administer formats. This agreement will further help us do so, as MGC’s experience in providing technical innovation in materials will serve as a strategic complement to our legacy of supporting the latest innovations in drug delivery for our customers.”
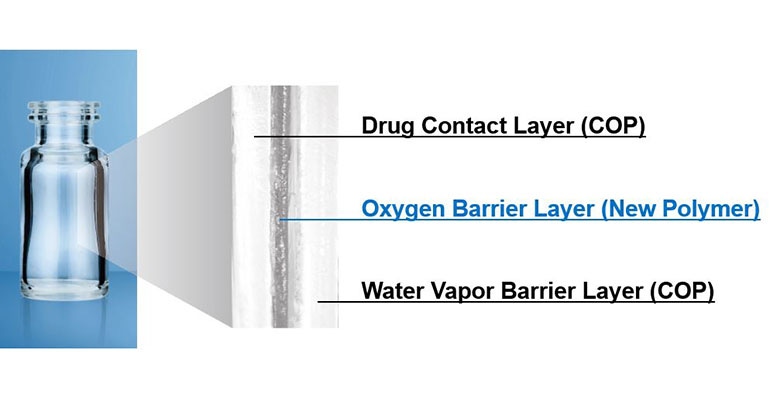
The Oxycapt multilayer structure is applied on silicon-free plastic syringe barrels that provide breakage resistance, an oxygen and vapor barrier, low protein adsorption, low extractables, and high UV barrier and pH stability, said MGC.
“Since launching Oxycapt, more and more pharmaceutical companies have been interested in applying it to their biologics or regenerative medicines such as gene/cell therapies,” said Ko Kedo, an executive officer of MGC’s R&D division. “As we have looked for a strategic partner to enhance presence of Oxycapt in the pharmaceutical industry, the partnership with BD will be an ideal solution for MGC. We believe this agreement will make it possible for customers to choose the best syringes for their drugs.”
Some pharmaceutical compounds based on mRNA, viral vector, or innovative antibody technologies may face challenges related to stability or integration with glass-based prefillable syringes, said BD. Through this partnership, BD and MGC will explore practical solutions for delivering newer biologic compounds.
Both companies noted that they share a commitment to advancing sustainability and reducing their carbon footprints.
You May Also Like


