New PEEK Formulation Improves Bone-Implant Fusion
The biomaterial expands Evonik’s portfolio of metal-free alternatives to titanium implants. It can be injection molded, milled, or extruded.
September 29, 2020

Specialty chemicals company Evonik has developed a new osteoconductive polyether ether ketone (PEEK) that improves bone-implant fusion. The material expands Evonik’s existing portfolio of biomaterials for implants in long-term contact with the body, offering a metal-free alternative to titanium implants. The new biomaterial, which is being marketed under the brand name VESTAKEEP Fusion, will be introduced at the virtual Eurospine conference on Oct. 6 to 9.
Biphasic calcium phosphate is added to the PEEK material to promote osteointegration, allowing bone cells to adhere to implants more quickly and promoting fusion at the boundary between the bone and implant. This, in turn, accelerates bone fusion and convalescence, said Evonik.
|
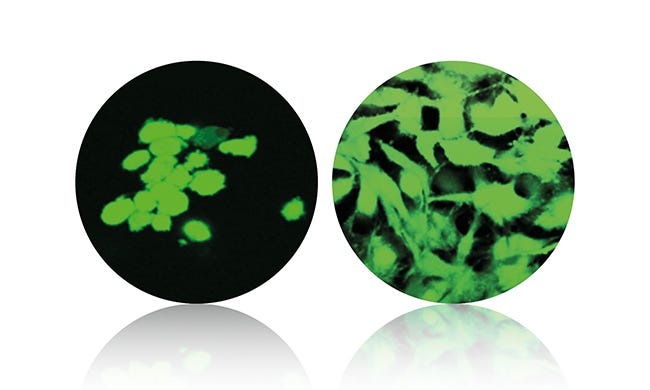
Adhesion of bone cells is shown on virgin VESTAKEEP material (left) and the new VESTAKEEP Fusion formulation. Image courtesy Evonik. |
The first biomaterial in Evonik’s new Fusion product line, VESTAKEEP iC4800 features similar mechanical properties to human bone. Like all other PEEK-based polymers for medical technology applications, the newly developed biomaterial does not produce any artifacts in imaging processes, but the additive provides a natural desired shadow for accurate placement and observation of the fusion process.
Development of customer-specific biomaterials
In addition to VESTAKEEP iC4800, Evonik offers the development of exclusive, customer-specific materials with bioactive properties in the new Fusion line. The VESTAKEEP Fusion Select products are developed from a library of established osteoconductive substances in collaboration with each customer.
VESTAKEEP Fusion is available in granulate form and as a semifinished stock-shape product. Like all previous PEEK products, it can be milled, compression molded, and extruded.
The new osteoconductive PEEK biomaterial can also be injection molded. It has been designed so that the functional additives are available on the surface without film formation. Evonik has collaborated with Samaplast, a Swiss company specializing in plastics engineering, to demonstrate and document the processing characteristics of VESTAKEEP Fusion in injection molding.
Evonik is currently studying the possibility of developing a 3D-printable filament with VESTAKEEP Fusion that could be processed via fused filament fabrication (FFF) technology.
About the Author(s)
You May Also Like