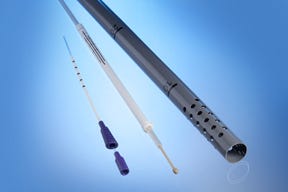
Pad-printed PEEK tubes from Putman Plastics
With an eye on the catheter market, medical tubing processor Putnam Plastics Corp. (Dayville, CT) has added pad-printed polyether etherketone (PEEK) and thermoset polyimide tubes to its portfolio. Plasma pre-treatment of the tubes ensures good print quality and involves little to no heat transfer, preserving the parts’ mechanical dimensions and properties. The company has developed its own pre-treatment technologies, including plasma pre-treatment, for its tubing.
December 11, 2009
With an eye on the catheter market, medical tubing processor Putnam Plastics Corp. (Dayville, CT) has added pad-printed polyether etherketone (PEEK) and thermoset polyimide tubes to its portfolio. Plasma pre-treatment of the tubes ensures good print quality and involves little to no heat transfer, preserving the parts’ mechanical dimensions and properties. The company has developed its own pre-treatment technologies, including plasma pre-treatment, for its tubing.
|
“Non-invasive vascular devices require integrity of the pad-printed graphics for the proper identification and use of the device by clinicians. Invasive devices require that particles of ink are not released from the device into the bloodstream which can lead to serious side effects or even death,” notes Byron Flagg, product manager–extrusion and component finishing, explaining the importance of adhesion of the printed graphics.
Putnam has made something of an art of secondary operations, even creating its own dedicated business unit, PPPC+, within Putnam that focuses specifically on reducing its customers’ need to outsource further. Not only printing, but other secondary operations such as grinding, hole drilling, mechanical flaring and more are done in-house. MPW covered other aspects of Putnam in an article earlier this year. —Matt Defosse
About the Author(s)
You May Also Like