In Extrusion as in Life, Follow the Science
Chemistry is the foundation of science. It may help you to better understand the extrusion process, and maybe other aspects of daily life, too.
March 13, 2022

Follow the science we are told, but it’s not so easy. Some of us learned science in college but don’t remember much, some in high school, and some not at all. We also want to follow our customers, who may know even less than we do. In the next couple of columns, I’m going to present summaries of the three basic sciences — chemistry, biology and physics — in simple language, condensed from the Simple Science booklets I’m working on now for wider distribution. I hope these articles will help you better understand the extrusion process, and maybe other aspects of daily living, too. Warning: many people are afraid of the logic of science. You can’t make deals with an atom, and there are no miracles. Numbers and probability matter. And the popular images are not always in accord.
Chemistry from a “simple science” perspective
Chemistry is basic to an understanding all science. And like all science, it cannot accept the impossible. There are still some unknowns, but what we know explains what we see.
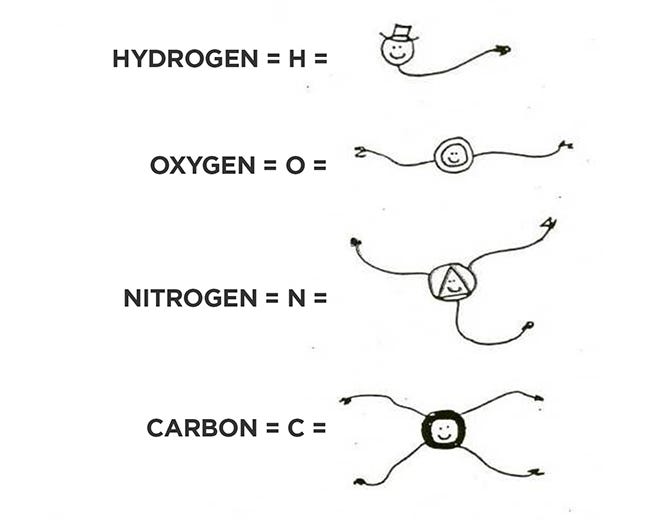
We start with atoms and molecules. An atom is the smallest particle of a substance that is still that substance. There are 92 such substances found in nature, the elements. (See diagram of four common elements below.) Almost all of them are solid at earth-surface temperatures; a few — bromine, mercury — are liquid; and a few more are gases — hydrogen, nitrogen, oxygen, fluorine, chlorine, plus the inert ones, helium, argon, neon, krypton, xenon, and radon.
|
Atoms are made up of smaller particles that may or may not carry an electric charge. We call them positive — the proton — and negative — the electron — and they attract each other. There is also the neutron with no charge at all, not quite a combination of an electron and proton.
Elements differ from one another in the number of these particles — a hydrogen atom is the smallest, with one electron and one proton. Uranium has the most, with 92 electrons, 92 protons and 146 neutrons (usually). We have also made artificial elements with more than 92 protons, but these decay quickly into the natural ones.
The protons and neutrons are in the atom’s nucleus. The electrons go around the nucleus like the planets around the sun or moons around a planet (like Jupiter, which has 79 moons). They repel each other (same charge) so they can’t get too close, and, therefore, they form fixed-number shells of 2, 8, 18, and 32.
Elements also differ in how many electrons are free to react. The inert gases don’t react because they already have complete electron shells and matching protons, but the other elements are reactive as they try to complete their shells even if that unbalances the atom. Hydrogen needs a second electron to make a shell of two, so it has one reactive site (like an arm, see diagram below). Oxygen needs two to make eight; nitrogen usually three, and carbon four.
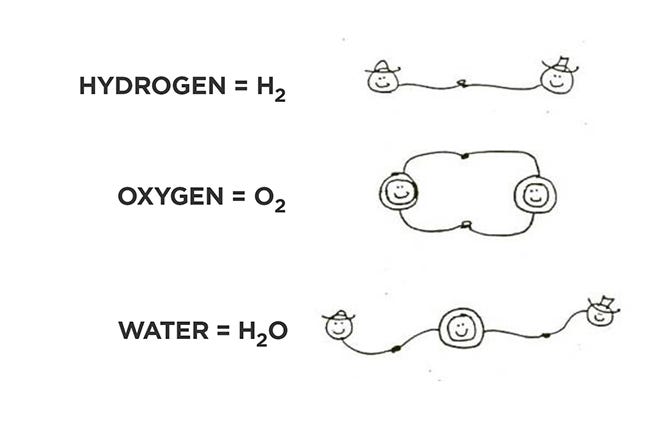
Molecules are the combinations of elements that we find in nature or can synthesize. They can be several elements (see diagram below), such as water or carbon dioxide, and some are not so simple — the blood component hemoglobin is a complex molecule with C, N, O, and H and iron atoms in the middle. Also, oxygen and the other reactive gases go around in pairs until they find something to react with: The oxygen in the air we breathe, for example, reacts with the nutrients we eat and digest.
Electra becomes electric
All these particles have mass, which is not the same as weight, as weight disappears in space (no gravity), but mass doesn’t. Protons and neutrons have much more mass — more than 2900 times — than electrons.
The word root electra comes from the Greek word for amber, a natural fossilized resin (polymer) made by prehistoric trees. It can be rubbed with another material like wool to generate static electricity, which is the collection of excess electrons on a surface. Electric power is based on movement of electrons through conductors (usually metals) and requires energy to push those electrons through the conductor’s resistance (electric cars are not magic). Elektra is also a character in Greek mythology (sister of Oedipus), important to understand, but not related to science.
Water is not an element, but a compound, a combination of two hydrogens with one oxygen (H2O), as shown in the sketch below. In the same way, one carbon can hold two oxygens to form carbon dioxide (CO2), which we and all animals (even bugs and fish) breathe out as a product of our metabolism, and plants take in as an ingredient of theirs. Trees are often angelized for this, but they give the CO2 back when their leaves drop off and decompose. Selective logic, the kind that says plastics are bad.
|
Many metals are elements — iron, copper, gold, lead, and aluminum — but some are alloys (zinc + copper = brass, tin + copper = bronze) and some are one element that has been processed, notably iron to steel. Some are found in nature as elements, but most are found as compounds (ores) and some like sodium and calcium are so reactive that they are never found uncombined. Good example: Sodium chloride = common salt.
The elements carbon and silicon are extremely abundant, usually combined with other elements. Carbon combinations are the basis of organic chemistry and form the molecules of living things as well as most plastics. The word organic here refers to the living things (animal and vegetable) as opposed to inorganic (mineral). This has nothing to do with organic agriculture (no chemical intervention) or the image of “natural” as opposed to synthetic. In discussions of climate change, the word carbon usually refers to carbon dioxide, not the element alone.
Air is not an element, but a mix of 78% nitrogen, 21% oxygen, 1% argon (inert), and some carbon dioxide and water (not inert and very important despite their low percentages).
If you’ve read this far, I suggest you go back and read it again, slow enough to digest. This is the foundation of chemistry without which we can’t understand the rest of Simple Science.
Building polymers
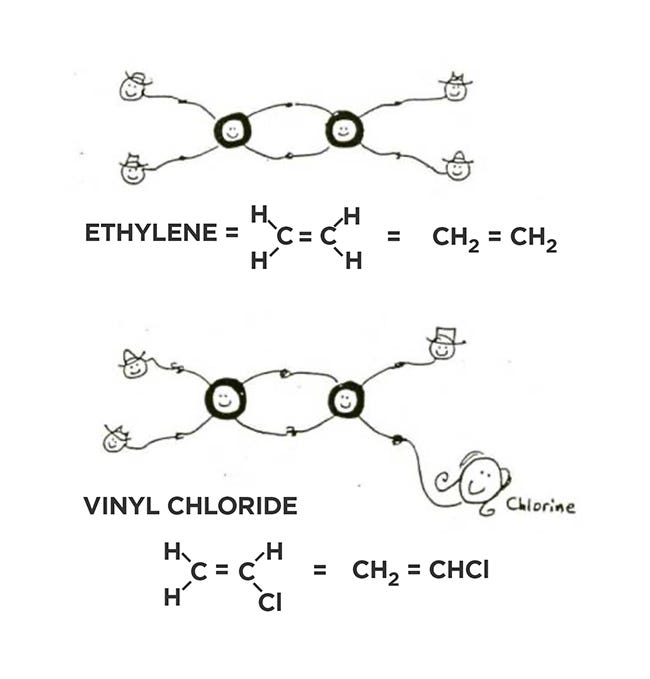
How do plastics fit into all this? Shown below are two molecules, ethylene and vinyl chloride, the building blocks (monomers) of two common plastics, polyethylene and PVC. You see a double bond, possible because a carbon atom has four “arms” and can hold another carbon with two of them, leaving the other two to hold hydrogens, oxygens, chlorines, or others.
|
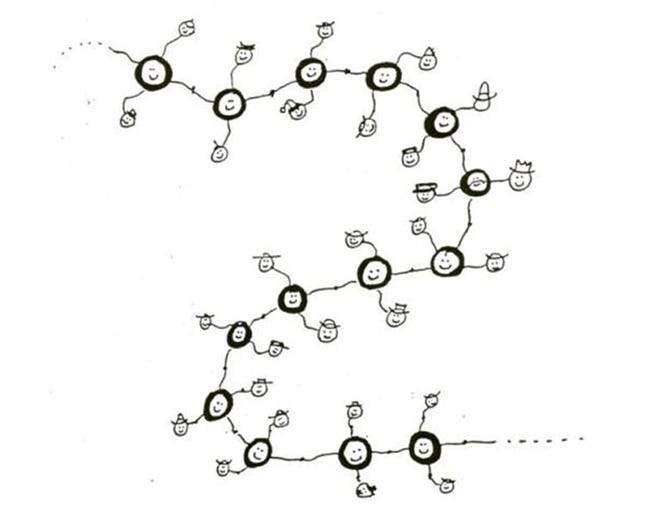
These double bonds are reactive, and one can break while the other remains. That way, long chains can be made if ethylene (or vinyl chloride) Is kept at the right temperature and pressure for enough time. Think of two people holding each other’s two hands. One hold can break and the free hands can grab onto free hands from other couples, and so on, to make the polyethylene chain shown below.
|
The molecules can get much more complicated, with branches and/or more than one monomer, and a few extrudable plastics, such as PET, PC, and nylons, are different, but still follow the same idea. Little molecules hook up to make big, long ones. Our genes are also strings of carbon-based molecules but that’s another (and very important) story.
If you’re dealing with the myth-understanding around BPA (bisphenol A), a monomer/building block for polycarbonate, you’ll need more detail. Ask any responsible polymer chemist. Write me if you can’t find one.

Allan Griff is a veteran extrusion engineer, starting out in tech service for a major resin supplier, and working on his own now for many years as a consultant, expert witness in law cases, and especially as an educator via webinars and seminars, both public and in-house, and now in his virtual version. He wrote Plastics Extrusion Technology, the first practical extrusion book in the United States, as well as the Plastics Extrusion Operating Manual, updated almost every year, and available in Spanish and French as well as English. Find out more on his website, www.griffex.com, or e-mail him at [email protected].
No live seminars planned in the near future, or maybe ever, as his virtual audiovisual seminar is even better than live, says Griff. No travel, no waiting for live dates, same PowerPoint slides but with audio explanations and a written guide. Watch at your own pace; group attendance is offered for a single price, including the right to ask questions and get thorough answers by e-mail. Call 301/758-7788 or e-mail [email protected] for more info.
About the Author(s)
You May Also Like