Weaker Polymer Bonds Produce Stronger Materials, Researchers Claim
A team of chemists from MIT and Duke University discovered they could increase the tear resistance of a polyacrylate elastomer by as much as 10 times by using a weaker cross-linker to bind the monomer chains.
June 27, 2023
We know that what doesn’t kill us makes us stronger — as Liam Neeson has taught us in almost every movie he’s ever made — but can weaker bonds in polymers actually make the material itself stronger? Yes, they can, claims a team of chemists from MIT and Duke University. They found that they could increase the tear resistance of a polyacrylate elastomer as much as 10-fold by using a weaker cross-linker to join the monomer chains.
“Polymer engineers know how to make materials tougher, but it invariably involves changing some other property of the material that you don’t want to change," said Stephen Craig, a professor of chemistry at Duke University, in an article on the MIT website. “Here, the toughness enhancement comes without any other significant change in physical properties — at least that we can measure — and it is brought about through the replacement of only a small fraction of the overall material,” said Craig, who is also a senior author of a paper describing the research in Science.
More tear-resistant car tires a possible application
Polyacrylate elastomer was selected for the study because it is easy to control, MIT professor Jeremiah Johnson and Shu Wang, an MIT postdoc who earned his PhD at Duke and is the lead author of the paper, told PlasticsToday. The material is also reproducible, enabling “relevant controls to rigorously test samples," they added, and it is a "commercially significant product used in adhesives and 3D printing." A particular application that caught researchers’ interest is car tires.
“If you could make a rubber tire 10 times more resistant to tearing, that could have a dramatic impact on the lifetime of the tire and on the amount of microplastic waste that breaks off,” said Johnson. The technology also may have applications in medical devices involving implanted cross-linked polymers, Johnson and Wang added.
“Side-chain cross-linked polymers are probably the most widely used type of polymer network,” Wang told PlasticsToday. “The concept [outlined] in our paper should work for all polymer networks that are side-chain cross linked.”
Building polymer networks
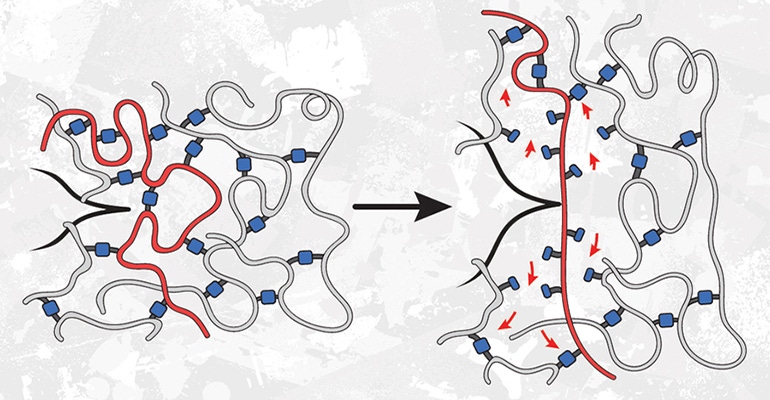
In polyacrylate elastomers, strands of acrylate hold together the polymer networks by linking molecules. These building blocks can be joined together in different ways to create materials with different properties, writes Anne Trafton of the MIT News Office in the release.
A so-called star polymer network is one architecture often used with these polymers: One building block is a star with four identical arms, and the other is a chain that acts as a linker. These linkers bind to the end of each arm of the stars, creating a network that resembles a volleyball net, explains Trafton.
In a 2021 study, researchers from MIT and Duke University teamed up to measure the strength of these polymers. As expected, they found that when weaker end-linkers were used to hold the polymer strands together, the material became weaker.
In a follow-up to that study, the researchers investigated a different type of polymer network in which polymer strands are cross-linked to other strands in random locations instead of being joined at the ends. This time, when the researchers used weaker linkers to join the acrylate building blocks together, they found that the material became much more resistant to tearing.
This occurs, the researchers believe, because the weaker bonds are randomly distributed as junctions between otherwise strong strands throughout the material, instead of being part of the ultimate strands themselves. When this material is stretched to the breaking point, any cracks propagating through the material try to avoid the stronger bonds and go through the weaker bonds instead. This means the crack has to break more bonds than it would if all of the bonds were the same strength.
The researchers have been exploring this phenomenon in commodity polymers, as well as rubber. The new studies are not published yet and the results cannot be released, but we “can say that the concept appears to work,” they told PlasticsToday.
The research is funded through a National Science Foundation center called the Center for the Chemistry of Molecularly Optimized Networks. The center’s mission is to study how the properties of the molecular components of polymer networks affect the physical behavior of the networks.
About the Author(s)
You May Also Like




