Medical-Grade TPE Film for Wound Dressings Alleviates Patient Discomfort
United Soft Plastics collaborated with a Denmark-based startup to develop an extruded TPE film for a medical device that improves patient comfort while providing bacterial protection in wound-dressing applications.
September 28, 2021
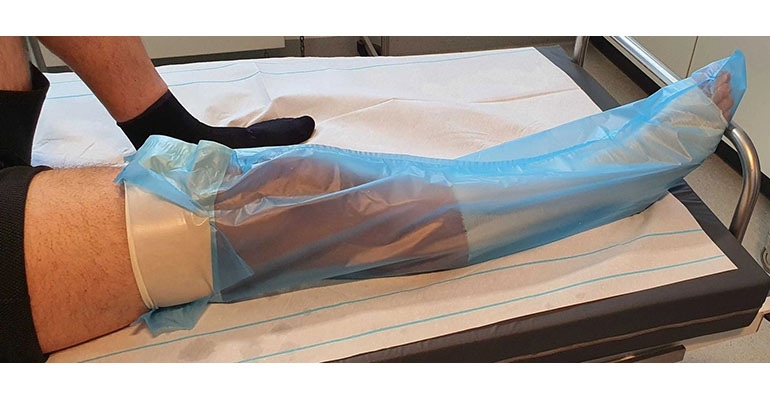
Thermoplastic elastomer (TPE) compounder United Soft Plastics Inc. has developed a custom extruded TPE film for wound-healing applications. The medical-grade TPE developed in conjunction with a Danish startup improves patient comfort while providing wound protection, according to United Soft Plastics (USP).
The area in proximity to wound dressings must be kept clean and dry before follow-up treatments or daily aftercare and protected from bacterial exposure. This is typically accomplished by covering the wound dressing with a medically protective film or protective bag (polybag), which is then affixed to the skin with an adhesive tape and sealed. Tearing off the tape to remove the protective film may subject the patient to pain in connection with micro-injuries to the topmost skin layer.
The custom medical-grade TPE, sold under the brand name Impervious, eliminates patient irritation and pain, said USP. The latex-free material does not have to be torn off because it is permanently welded to the polybag as a stretchable, flexible, and conical sealing strip. Adhesive tape is not used in this construction.
USP was skeptical that the project was feasible when the startup initially pitched the idea. “We were not sure whether we would actually be able to develop a medical TPE grade that combines a multitude of partly contrary performance properties along with the need to meet strict customer and medical regulatory requirements,” said Michael Bodmann, USP General Manager-Europe.
The TPE grade, currently in pre-commercialization, exhibits a low 25 to 35 Shore A hardness, high tear strength in connection with demanding transverse and longitudinal strain loads, and very thin 0.2- to 0.4-mm wall thickness. The Danish company also addressed manufacturing issues so the medical-grade TPE could be produced in a cost-effective manner, said Bodmann.
The result of a year-long joint development, Impervious is now undergoing clinical trials. After initial commercialization in Europe, the new TPE film will be made available globally.
About the Author(s)
You May Also Like


