Medical Plastics 101: Cyclic Olefin Copolymer Fulfills Complex Medtech Performance Requirements
A balance of properties, including purity, clarity, sterilizability, and chemical resistance, drive use of the polymer in applications ranging from diagnostic products and wearable insulin devices to primary drug packaging.
February 2, 2021
Timothy Kneale
Cyclic olefin copolymer (COC) is a unique member of the polyolefin family, which also includes high-volume materials like polyethylene (PE) and polypropylene (PP). It was introduced a few decades ago and has become a widely used material in medical and packaging applications. The extremely high purity and unique properties of COC have made it the dominant material in advanced diagnostic and microfluidic applications. The material’s use in primary drug packaging — in the form of prefilled syringes, vials, blisters, pouches, and lightweight wearables — has become increasingly common as newer pharmaceutical molecules and formulations trend toward more chemical sensitivity to lower-purity plastics and glass. PE and PP films use COC as a performance enhancer to simplify films and compete with more complex, less recyclable structures.
Unique balance of properties
Part of the appeal of COC is its glass-like clarity, which matches or exceeds traditional glass substitutes like polycarbonate (PC) and polymethyl methacrylate (PMMA). Importantly for medical uses, COC is sterilizable by all standard methods including steam, EtO, gamma, and hydrogen peroxide. It has the best UV transmission of any polymer, which is key for many diagnostic analyses. Grades with heat resistance up to 170°C are available, easily withstanding polymerase chain reaction (PCR) and steam sterilization conditions. Another powerful advantage of COC is its resistance to aggressive polar chemicals. It is highly tolerant of acids, bases, alcohols, and more. The resin also provides one of the best barriers to moisture of any plastic.
The advantages of COC in medical design are compelling. The resin is capable of reproducing incredibly fine detail via injection molding, even at the submicron level. It also has exceptional dimensional stability and low shrinkage, enabling volumetric precision most competitive resins cannot match.
|
Sandstone Diagnostics selected TOPAS COC from Polyplastics for its male fertility management system because of the material's low moisture vapor transmission rate, decreased water absorption, biocompatibility, and low moisture vapor transmission rate. |
Perhaps the leading attraction of COC is its extremely high purity and inertness. Its low leachables and extractables make it ideal for direct drug contact, while the virtual absence of ions preserves potency of sensitive formulations. It is widely used in the most challenging drug-delivery and packaging applications where even medical glass does not work. In diagnostic and microfluidic applications, the same characteristics ensure that the material being analyzed or processed remains uncontaminated, maximizing analytical accuracy and product purity. As one might expect, regulatory compliance is a strong advantage for COC, with USP Class VI and ISO 10993 compliance including biocompatibility, USP 661.1, and FDA drug and device master files.
COVID-19 related applications emerge
The COVID-19 pandemic has created a surge of interest in COC for diagnostic disposables and for vaccine and therapeutic packaging. COC has been a longtime material solution for medical syringes and vials, as well as disposable diagnostic containers, markets that have been dominated by borosilicate glass.
COC is being used in COVID-19 testing applications, and developments are ongoing for its use in new vaccines and therapies. With a range of new tests and therapies under consideration, companies are using the purest, most inert products for their developments. The interest in COC has been further elevated by increased concern in the pharmaceutical industry of a shortage of glass vials for COVID-19 vaccines.
COC could serve to alleviate a potential shortage of borosilicate glass used to make vaccine vials. While inexpensive borosilicate glass meets today’s industry needs, there are emerging drugs and therapies that are incompatible with glass. In particular, COC could fill a vital role as more biotech-derived active ingredients emerge.
The material’s extremely low leachables and extractables, along with its non-polar, low-reactivity surface and broad global regulatory compliance make it ideal for diagnostic disposables, and for packaging of vaccines and therapeutic agents. COC is currently commercial in many medical applications at leading global healthcare companies. The material provides ultra-high purity and its inert nature prevents interference with reactions and analyses. Additional key features include superior optical performance (91% clarity) along with excellent UV transparency and low birefringence.
Range of healthcare applications
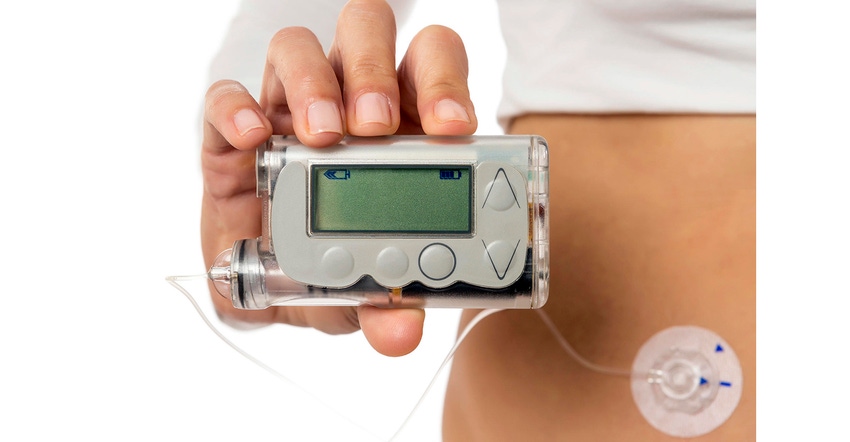
COC is seeing strong market growth in the medical sector as its advantages become more widely known, and as supply continues to increase. Prefilled syringes are a notable success story, building on the outstanding purity and resulting drug stability delivered by COC syringes. Another booming application is wearable insulin delivery devices, where the purity and dimensional stability of COC result in high potency and precise medication control.
Various devices use COC for chemical resistance, with bone cement mixers being a specialty, while there has been a jump in the number of medical devices using COC to upgrade the performance of PE and PP via blending, thereby replacing more expensive polymers. On the diagnostic front, COC disposables used in disease testing are increasingly popular, largely replacing PP and polystyrene (PS) when more precise analyses are required.
Microfluidic applications have evolved and benefitted from several innovations throughout the years. New designs have expanded use from simple microanalysis performed in the lab to the rapidly growing area of point-of-care (POC) diagnostics. With these advances, the choice of materials used to fabricate biochips, cartridges, and other microfluidic components also continues to evolve. Glass and polymers like silicone were mainstays for many years. More recently, cyclic olefin copolymer has emerged as a highly useful and attractive microfluidic material, offering high optical and UV clarity, low water absorption, exceptional moisture barrier, and excellent resistance to chemicals, including leading organic solvents used in chemical analysis.
COC’s unique properties make it an excellent material for the design and manufacture of microfluidic parts used in analytical systems, research, and biomedical devices. COC can be used to replicate features including microchannels with fabrication processes such as hot embossing for low to medium throughput, and injection molding for faster production of high-quantity detailed parts.
COC lifts performance of polyolefin blends
Cyclic olefin copolymer is finding increased utility as a performance enhancer in polyolefin-based film packaging. In polyolefin blends, COC offers a major performance upgrade, providing enhanced modulus, greater thermal resistance, and increased barrier for thermoformed articles such as trays, along with bags and pouches. These blends are easily processed on conventional cast and blown film processing equipment within standard operating parameters for polyolefins.
COC is based on the polymerization of ethylene and norbornene using metallocene catalysts. The material has a glass transition temperature (Tg) ranging from 65o to 180o C as a function of comonomer ratios. As an amorphous polymer, COC does not have a crystalline melting point and instead starts to soften above Tg, becoming increasingly fluid as temperatures are increased. This colorless, crystal-clear material possesses high modulus and offers strong compatibility with conventional polyethylene grades. Compatibility is best with linear polyethylene products such as LLDPE and HDPE, and is acceptable for LDPE. Processors have for many years reprocessed COC/PE trim and scrap directly back into production. Recognizing the increasing importance of recyclability, recent testing has shown that COC is compatible with PE and PP post-consumer recycling streams in Europe, and with PE for containers in the United States. More testing is underway, including the newest US PE film protocol.
As a blend component, COC is most commonly used to enhance modulus and allow thickness reduction of a monolayer film. With a modulus in the 300,000 psi range, as little as 10% of COC added to LLDPE will double or triple its modulus while maintaining a low haze level. In either a simple monolayer film or as part of a co-extruded film, a LLDPE/COC blend often allows a thinner film to provide the same performance at a reduced area-based cost, thus offering important sustainability benefits.
When higher Tg grades of COC are utilized, these modulus improvements are maintained up to temperatures approaching the Tg of the COC, improving hot-fill performance and elevating temperature capability. Another important point is that the addition of COC to LLDPE can significantly reduce Elmendorf tear values, especially in the machine direction, even as it improves the puncture resistance of the film. COC is utilized commercially as a controlled, linear-tear additive. Additionally, many olefin/COC films are used commercially in thermoforming applications where COC’s amorphous nature improves the uniformity of the forming process, allowing improved thickness control in deep-draw (>10 cm) sections and corners.
|
COC can be blended with polyolefin for high-clarity barrier films and in multilayer constructions to enhance performance. |
Multilayer polyolefin films are enhanced
Cyclic olefin copolymer also enables higher performance in multilayer polyolefin packaging films for many applications. COC/LLDPE multilayer films have improved formability, optics, toughness, and puncture resistance versus similar EVA/ionomer co-extruded films at a comparable cost basis. Engineered COC/LLDPE multilayer films can also compare favorably with many nylon-based structures while eliminating the need for adhesive tie layers. Removing tie layers and non-olefin resins simplifies production logistics and permits the use of recycled scrap film because of the compatibility between COC and polyethylene.
COC also increasingly is being used to produce high-clarity barrier films for packaging. COC has moisture-barrier capabilities, which are four to five times better than LDPE. Blends that are composed of more than 70% COC typically provide up to 90% of the barrier performance of neat COC and – unlike HDPE — maintain low haze. While COC can enhance barrier performance as a blend component, the best barrier improvement is realized when COC is utilized in a discrete layer. COC also typically provides five to 10 times better barrier resistance to aromas and fragrances than LLDPE. Although COC does not offer high gas barrier, its performance is still significantly better than polyethylene and can be used to tailor permeation to meet the specific oxygen, nitrogen, and carbon dioxide barrier resistance required in packaging.
Opportunities and trade offs
COC applications tend to be in areas where traditional materials cannot handle one or more performance requirements. While PC is widely used in medical applications and offers impact properties and ductility superior to COC, it sometimes cannot meet the purity or thermal demands of medical applications. On the other hand, PMMA’s impact properties are similar to COC, but acrylic’s heat resistance is markedly inferior to COC. In the polyolefin world, COC rarely competes with inexpensive PE and PP, but is often combined with these materials (via blending or multilayering) to enhance properties such as heat and chemical resistance, dimensional stability, barrier, thermoformability, and more. One area where COC is limited is contact with fats, oils, and fuels. These nonpolar materials may permeate COC and mar the surface.
Unlike other polyolefins, COC is an amorphous polymer. This gives it processing characteristics similar to widely used medical resins such as PC and PMMA. It can be injection molded, and also makes its way into extruded medical film products such as blister packaging and barrier pouches. Many leading medical injection molders are fully experienced with COC, as are many top film manufacturers.
COC is a chemical cousin of another amorphous polyolefin, known as COP (cyclic olefin polymer). This resin shares many high-performance attributes with COC and is used in many of the same applications, with each having minor advantages in certain performance categories. However, COP is manufactured via a more complex process than COC, which often results in COC being a more cost-effective choice.
Conclusion
COC is a vital addition to the arsenal of medical and packaging materials. It enables better medication stability and unmatched disease diagnostic capability, while delivering improved performance and recyclability to plastic films. Continued strong growth is expected, as this material fills complex performance requirements and enables many of the growing trends in the medical industry, including drug complexity, point-of-care testing, and wearable devices. The need for efficient, sustainable packaging will continue to drive growth in packaging markets.
About the author
Timothy Kneale is President of TOPAS Americas, Polyplastics USA. Trained as a chemical engineer, Kneale has held a variety of plastics industry product development and technical leadership roles. Since 2008, he has led the TOPAS COC business in the Americas. Polyplastics Co., Ltd., via its subsidiary TOPAS Advanced Polymers GmbH, is a leading manufacturer of COC, marketed under the brand name TOPAS. For more information, visit topas.com or contact Kneale at [email protected]
You May Also Like




